Dr. Amit V. Janbandhu & Dr. Sanjay Singhal
1. Introduction
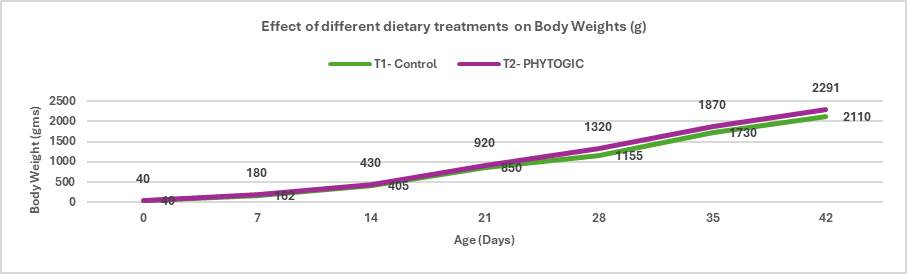
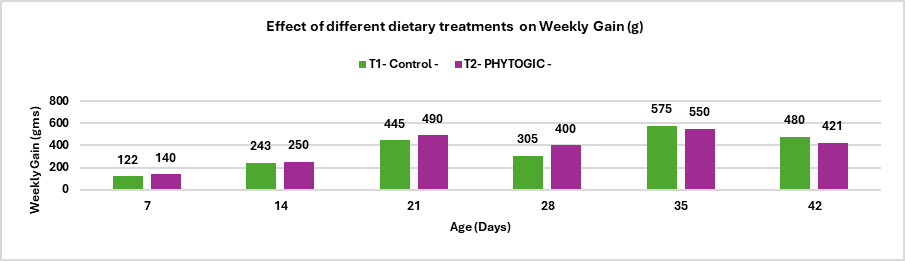
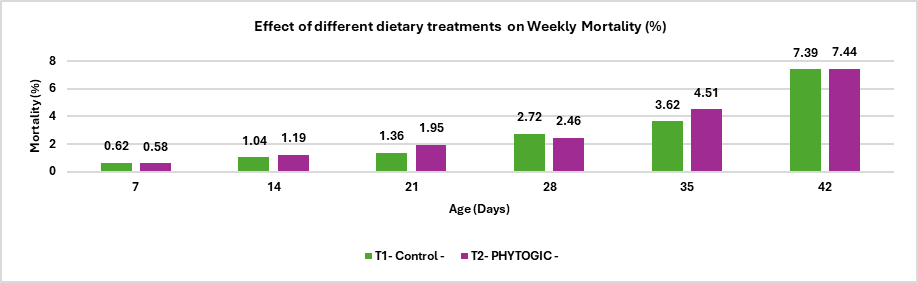
Animal fats and vegetable oils are commonly incorporated into poultry diets due to their high energy density, which supports optimal growth performance (Blanch et al., 1996). Dietary fats and oils provide approximately 2.25 times more metabolizable energy than carbohydrates and serve as important sources of essential fatty acids and fat‑soluble vitamins. In recent years, rising feed costs have increased interest in maximizing dietary fat utilization to enhance energy concentration and meet the demands of high‑performing birds. However, fat absorption efficiency is age‑dependent; young broilers exhibit a physiological limitation in lipid digestion and absorption, which improves progressively with advancing age (Kussaibati et al., 1982)
Enzymatic hydrolysis of lipids (oils and fats) produces fatty acids (FA) which are water insoluble. FA passes through the liquid phase of the small intestine and, after aggregating to form micelles, are absorbed as hydrophobic components. This process is naturally mediated by endogenous emulsifiers, such as bile salts. The assimilation of dietary fats in young birds is poor because they have a limited capacity to produce and secrete bile salts and lipase until their gastrointestinal tract matures at 10-14 days of age (Noy and Sklan,1998).
This review evaluates the role and application of exogenous emulsifiers, such as Lipifier-DS from Stallen South Asia Pvt Ltd., a multi-component emulsifier and absorption accelerator, in nutrient-dense broiler diets to maximize the growth potential of modern poultry genetics.
2. Digestibility problems in young chicks
Young birds exhibit limited fat absorption due to low endogenous lipase activity, reduced bile secretion, and poor emulsification capacity; however, these functions improve with age and adapt to higher levels of unsaturated fatty acids (Meng et al., 2004). Consequently, immature digestive systems are unable to efficiently form mixed micelles in the intestinal lumen, leading to reduced fat digestion and nutrient absorption (Leeson and Atteh, 1995). Age‑related differences in metabolisable energy (ME) utilization and growth performance have therefore stimulated interest in the use of exogenous emulsifiers to enhance fat utilisation in young birds (Roy et al., 2010). High dietary inclusion of saturated fats in broiler rations can lead to excessive visceral and carcass fat deposition, reduced vitamin A and E availability, and compromised meat quality (Chae et al., 2006).
3. Energy efficiency of emulsifiers in High‑Performance Broiler Diets
Energy is a major cost component factor in diets of high-performance animals, such as broilers. Emulsifiers can be used to improve fat digestibility and energy efficiency. As a result, lower energy diets can be formulated for birds whilst maintaining the same performance, leading to lower feed cost and more economical and sustainable production. Emulsifiers facilitate the formation of emulsion droplets, which lowers the surface tension (Ashraf, 2007), stimulates the formation of micelles, causes high levels of monoglycerides in the intestine and facilitates the nutrient transport through the membrane (Melegy et al., 2010). Emulsifier supplementation has been shown to improve feed efficiency, lipid absorption, and blood lipid profiles, although its effects on growth performance and carcass traits are inconsistent (Udomprasert and Rukkwamsuk, 2006).
4.Classification and Functional Role of Exogenous Emulsifiers in Broilers
Exogenous emulsifiers used in animal nutrition are broadly classified as natural or synthetic, with natural emulsifiers—including bile salts, phospholipids, and dietary sources such as soy lecithin—and synthetic emulsifiers comprising chemically modified molecules such as lysolecithin or lysophosphatidylcholine (Zhang et al., 2011). By modifying hydrophobic interfaces and promoting mixed‑micelle formation, these emulsifiers enhance fat digestibility, particularly in young birds with limited endogenous emulsification capacity (Al‑Marzooqi and Leeson, 1999). Soy lecithin remains the most extensively validated natural emulsifier in poultry, improving fat utilization, growth performance, and serum lipid profile while supplying choline to prevent perosis (Polin, 1980; Siyal et al., 2017; Schaible, 1970). Synthetic emulsifiers, including polyethylene glycol mono‑ and dioleates and sodium stearoyl‑2‑lactylate, have also been shown to improve growth performance and the utilization efficiency of fat, protein, and metabolizable energy, although some synthetic polyoxyethylene glycol emulsifiers exhibit lower in vivo efficiency compared with bile salts (Frobish et al., 1969; Roy et al., 2010).
5. Emulsifying agents
An emulsifying agent stabilizes an emulsion by reducing interfacial tension between immiscible phases such as oil and water, thereby preventing droplet coalescence. Emulsifiers possess both hydrophilic and lipophilic moieties, enabling their adsorption at the oil–water interface and stabilization of dispersed fat droplets. Efficient fat emulsification is a prerequisite for lipid digestion in the gastrointestinal tract and is influenced by fatty acid chain length, triglyceride structure, and degree of saturation (Gu and Li, 2003). Exogenous emulsifiers enhance lipid utilization, particularly of animal fats, and partially compensate for limited bile production and enterohepatic recirculation in young birds. Although bile salts and monoglycerides function as natural emulsifiers, their emulsification capacity is insufficient in young birds, resulting in poor fat digestibility. Furthermore, saturated and free fatty acids exhibit a lower capacity for micelle formation than long‑chain unsaturated fatty acids, further limiting lipid digestion efficiency.
Table 1. Available Emulsifiers Used in the Poultry Industry
| Category | Emulsifier | Major Effects | References |
| Natural emulsifiers | Soy lecithin | Improved growth performance; increased HDL; reduced triglycerides, insulin (INS), thyroid‑stimulating hormone (TSH), total cholesterol (TC), and LDL levels | Siyal et al., 2017; Huang et al., 2007 |
| Milk‑derived casein | Increased weight gain, improved feed conversion ratio (FCR), enhanced pancreatic lipase activity, improved ether extract digestibility, with no effect on carcass traits or serum cholesterol | Guerreiro et al., 2011 | |
| Lysophosphatidylcholine / Lysolecithin | Improved weight gain and FCR; increased digestibility of total tract apparent digestibility (CTTAD) of C16:0, C18:2, and C18:3n‑3 fatty acids; improved carcass quality and dressing percentage | Melegy et al., 2010; Zhang et al., 2011; Azman and Ciftci, 2004; Ashraf, 1995 | |
| Bile salts | Increased relative organ weights; significantly improved body weight gain (BWG); decreased plasma cholesterol; increased metabolisable energy; enhanced digestive enzyme activity in the intestinal tract | Abd‑El‑Rauof, 1995; Alzawqari et al., 2010; Gomez and Polin, 1976; Kussaibati et al., 1982; Zhong and Xiang, 2008 | |
| Synthetic emulsifiers | Glycerol polyethylene glycol ricinoleate (E 484) | Improved performance; increased intake and utilization efficiency of fat, crude protein (CP), and metabolisable energy (ME) | Roy et al., 2010 |
| Lipex® (commercial emulsifier) | Increased body weight gain and liver weight | Yordan et al., 2013 | |
| Sodium stearoyl‑2‑lactylate (SSL) | Improved body weight and relative organ weights | Flores et al., 2007; Chronakis et al., 2004 |
6. Principles of Emulsification
Emulsifiers lower the interfacial energy between the two immiscible liquids, thereby helping the formation of an emulsion. For an emulsifier to be effective at reducing droplet size and stabilising an emulsion, it needs to be located at the interface. The emulsifier must not be too soluble in either phase, otherwise it will migrate to that phase. If the emulsifier migrates away from the interface, the emulsion is destabilised.
An emulsion is most accurately defined as a dispersion of liquid droplets in a second immiscible liquid. Temporary emulsions may be formed by mixing/agitating the two normally immiscible liquids; however, the stability of temporary emulsions produced in this way is poor. Emulsifiers are surface active materials (surfactants) that are used to assist in the formation of an emulsion and to stabilise the emulsion.
Fig.1. Below is a simplistic view of an emulsion particle, protected by surfactant molecules partitioned at the interface of the internal and external phase.
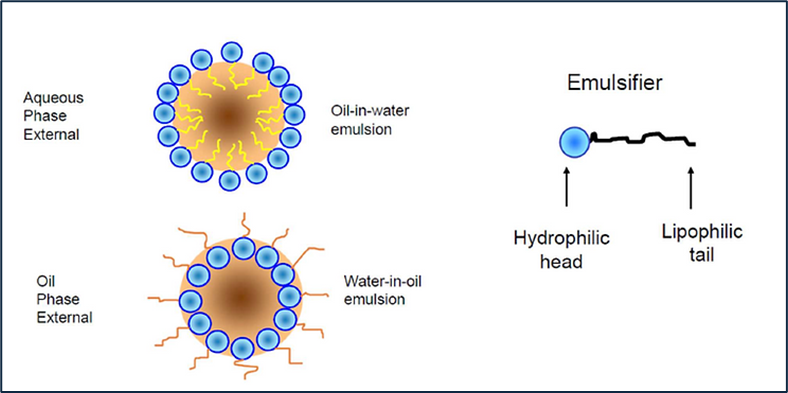
There are several different types of emulsions. They are loosely described by their phase relationship and/or by their appearance:
- Oil-in-Water
- Water-in-Oil
- Multiple emulsions
- Macro-emulsions
- Micro-emulsions
The appearance of the emulsion is dependent upon the particle size of the discontinuous phase.
Table.2. Particle size is listed in nanometers (nm)
| Particle size | Appearance |
| > 1 | White |
| 0.1 -1.0 | Blue White |
| 0.05 – 0.1 | Translucent |
| < 0.05 | Transparent |
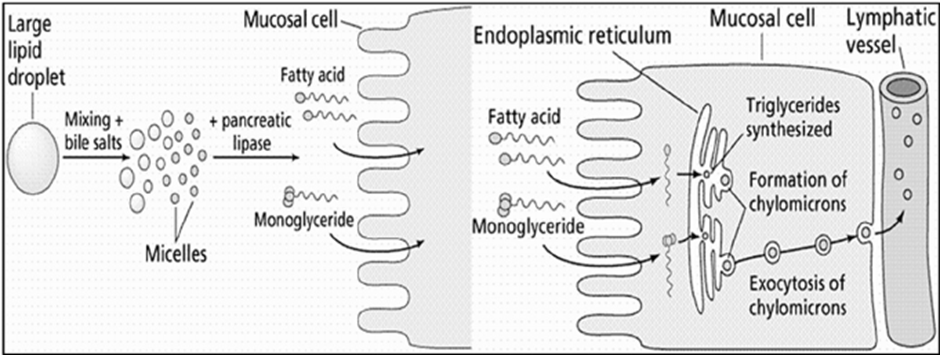
7. Mechanism of Action of Lipid Digestion and Absorption
- Emulsification of dietary fat: Large lipid droplets entering the intestinal lumen are emulsified by bile salts, reducing surface tension and breaking them into smaller droplets. This process increases the surface area available for enzymatic action.
- Enzymatic hydrolysis of lipids: Pancreatic lipase acts on the emulsified fat droplets, hydrolyzing triglycerides into free fatty acids and monoglycerides.
- Micelle formation: The released fatty acids and monoglycerides associate with bile salts to form mixed micelles, which are water-soluble and capable of diffusing through the intestinal lumen.
- Transport across the mucosal membrane: Micelles deliver fatty acids and monoglycerides to the brush border of intestinal mucosal cells, where these lipids diffuse across the cell membrane into the enterocytes.
- Re-esterification within the enterocyte: Inside the mucosal cells, fatty acids and monoglycerides are transported to the endoplasmic reticulum, where they are re-esterified to form triglycerides.
- Chylomicron formation: Newly synthesized triglycerides are packaged with phospholipids, cholesterol, and apoproteins to form chylomicrons.
- Exocytosis and lymphatic transport: Chylomicrons are transported to the basolateral membrane of the enterocyte and released by exocytosis into the lymphatic vessels, from where they enter systemic circulation.
Fig.2. General schematic of lipid digestion and absorption
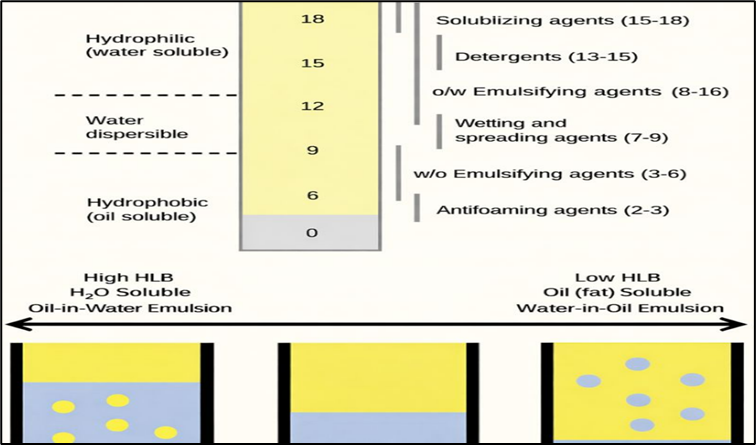
8. Hydrophilic-lipophilic balance
The combination of hydrophilic and lipophilic characteristics in one molecule gives it the distinctive property that the emulsifier can dissolve in fat as well as in water, and can aid in mixing the two fractions. The key indicator for selecting an emulsifier is hydrophilic-lipophilic balance (HLB), ranging from 0 to 20 which reveals the degree of fat or water solubility. Lower HLB indicates a more lipophilic or fat-soluble emulsifier. On the other hand, higher HLB indicates a more water soluble or hydrophilic emulsifier. Ideally, the emulsifier should be soluble in the continuous phase as the Bancroft rule states (1912). For the soluble condition known as the ‘fat-rich environment’ mixed in a small amount of water, an emulsifier with a lower HLB is advised, and vice versa. Because of birds consume water 1.5-2 times more than feed; the diet should contain a small amount of fat and the water amount should exceed fat in digestive tract. In this situation, a high HLB is more appropriate.
Fig. 3. HLB scale and its influence on surfactant functionality and emulsion types (Al-Yami et al. 2018)
9. Lipifier‑DS: –
Lipifier‑DS contains a blend of hydrolysed phospholipids—including LPC, LPE, LPI, LPA, other lysophospholipids—along with glyceryl polyethylene glycol ricinoleate coating.
a) Mechanism of Action of Lipifier-DS
Lipifier-DS function based on their solubility. If the emulsifier is more soluble in water, it forms an oil-in-water (O/W) emulsion, ideal for fat digestion in poultry. Conversely, if it is more soluble in oil, it forms a water-in-oil (W/O) emulsion. The emulsification process helps break down fat droplets into smaller micelle particles that remain dispersed in the water phase, allowing digestive enzymes to act more efficiently and resulting in availability of extra metabolizable energy to the birds. Exogenous emulsifiers are molecular surfactants with both hydrophobic and hydrophilic properties. The hydrophobic end with fatty acids is directed to the oil phase, while the hydrophilic end with sucrose, glycol, glycerol, sorbitol or polyglycerol is directed to the aqueous phase, forming a “molecular bridge” by decreasing surface tension that inhibits the coalescing of hydrolysed lipid droplets into large molecules.
b) Key Features of Lipifier-DS
- It contains Glyceryl Polyethylene Glycol Ricinoleate (PEGR) coating for stable and efficient emulsification.
- Lipifier-DS has optimized HLB (Hydrophilic–Lipophilic Balance) value of 9–12, ensuring effective emulsification and superior fat utilization.
- Highly efficient energy contributor — 250 g of Lipifier-DS provides 40,000 Kcal/kg, equivalent to approximately 4.25 kg of soybean oil. Enhances dietary energy efficiency while reducing dependence on added oil sources.
c) Benefits of Lipifier-DS
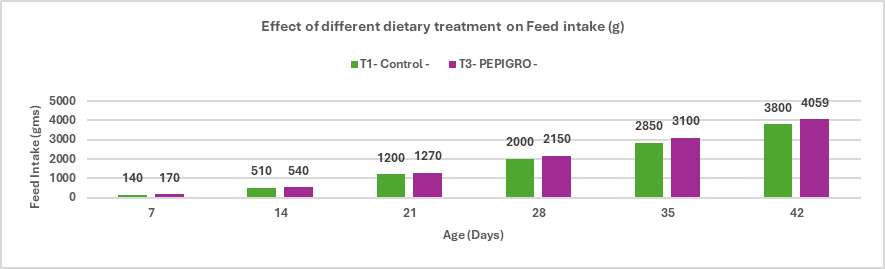
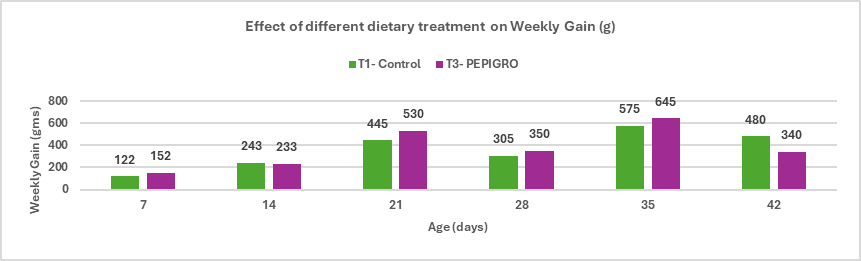
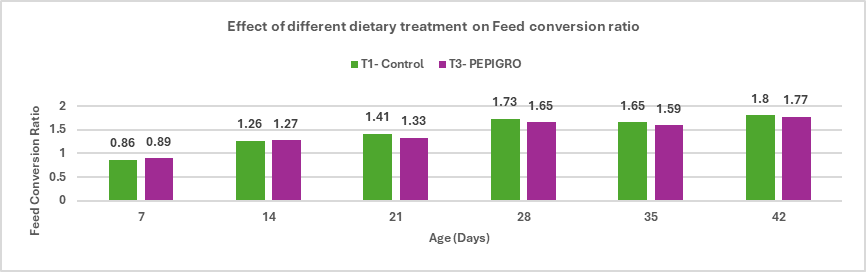
- Enhances growth performance and improves feed conversion ratio (FCR) in broilers.
- Improves egg production performance and increases egg size in layers.
- Enhances digestion and absorption of fats and fat-soluble vitamins.
- Promotes better absorption of essential nutrients.
- Ensures effective emulsification by forming a stable emulsion with PEGR coating.
- Improves fat digestibility and maximizes energy availability.
- Extracts greater nutritional value from feed, improving overall nutrient utilization.
10. Conclusion:
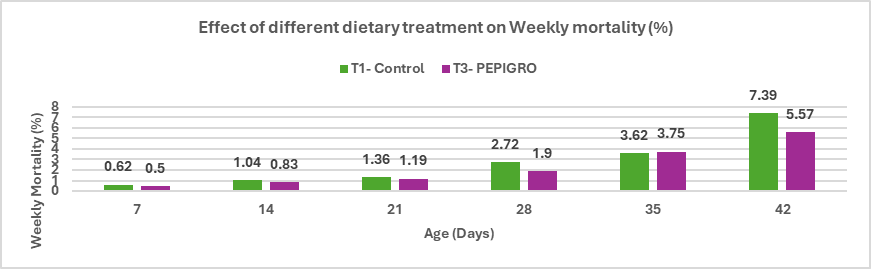
Stallen South Asia Pvt. Ltd.’s has Lipifier-DS is an effective exogenous emulsifier that enhances fat utilization and nutrient absorption in poultry diets. By promoting efficient emulsification through a stable PEGR coating, it improves energy availability and feed efficiency, supporting better growth and FCR in broilers, enhanced egg performance in layers, and overall higher production efficiency.
References
BLANCH, A., BARROETA, C., BAUCELLS, M.D., SERRANO, X. and PUCHAL, F. (1996), Utilisation of different fats and oils by adult chickens as a source of energy, lipid and fatty acids. Animal Feed Science and Technology 61: 335-342.
NOY, Y. and SKLAN, D. (1998) Metabolic responses to early nutrition. Journal of Applied Poultry Research 7: 437-451.
Kussaibati R, Guillaume J and Leclercq B. 1982. The effects of age, dietary fat and bile salts, and feeding rate on apparent and true metabolisable energy values in chickens. British Poultry Science 23(5): 393–403.
Meng X, Slominski B A and Guenter W. 2004. The effect of fat type, carbohydrase, and lipase addition on growth performance and nutrient utilization of young broilers fed wheat-based diets. Poultry Science 83(10): 1718–27.
LEESON, S. and ATTEH, J.O. (1995) Utilisation of fats and fatty acids by turkey poults. Poultry Science 74: 2003-2010.
ROY, A.S., HALDAR, S., MONDAL, T. and GHOSH, K. (2010) Effects of supplemental exogenous emulsifier on performance, nutrient metabolism, and serum lipid profile in broiler chickens. Veterinary Medicine International: Art. ID 262604.
ASHRAF, M. (2007) Use of Emulsifiers in High Fat Level Diets of Broilers. Doct thesis, Dept Animal Production, Faculty of Agriculture, Al Azhar University, Cairo, Egypt, 235, 2007.
MELEGY, T., KHALED, N., EL-BANA, R. and ABDELLATIF, H. (2010) Dietary fortification of a natural biosurfactant, lysolecithin in broiler. African Journal of Agriculture Research 5: 2886-2892.
UDOMPRASERT, P. and RUKKWAMSUK, T. (2006) Effect of an exogenous emulsifier on growth performance in weanling pigs. Kasetysart Journal of Natural Science 40: 652-656.
Zhang B, Haitao L, Zhao D, Guoand Y and Barri A. 2011. Effect of fat type and lysophosphatidylcholine addition to broiler diets on performance, apparent digestibility of fatty acids and apparent metabolisable energy content. Feed Science and Technology 163: 177–84.
AL-MARZOOQI, W. and LEESON, S. (1999) Evaluation of dietary supplements of lipase, detergent and crude porcine pancreas on fat utilisation by young broiler chicks. Poultry Science 78: 1561-1566.
Siyal F A, Babazadeh D, Wang C, Arain M A, Saeed M, Ayasan T, Zhang L and Wang T. 2017. Emulsifiers in poultry industry- A review. World Poultry Science Journal 73: 1–6.
Schaible P J. 1970. Poultry: Feeds and Nutrition. 2nd Edn., The AVI Publishing Co. USA.
Polin D and Hussein T H. 1982. The effect of bile acid on lipid and nitrogen retention, carcass composition, and dietary metabolizable energy in very young chicks. Poultry Science 61(8): 1697–1707.
FROBISH, L.T., HAYS, V.W., SPEER, V.C. and EWAN, R.C. (1969) Effect of diet form and emulsifying agents on fat utilisation by young pigs. Journal of Animal Science 29: 320-324.
CHAE, B.J., LOHAKARE, J.D. and CHOI, J.Y. (2006) Effects of incremental levels of α-tocopherol acetate on performance, nutrient digestibility and meat quality of commercial broilers. Asian-Australian Journal of Animal Sciences 19: 203-208.
GU, X. and LI, D. (2003) Fat nutrition and metabolism in piglets: A review. Animal Feed Science and Technology 109: 151-170.
BANCROFT, W.D. (1912) The theory of emulsification VI. The Journal of Physical Chemistry 17: 501-519.