Dr. Amit V. Janbandhu & Dr. Sanjay Singhal
1. Introduction
Global demand for poultry meat and eggs is rising with population growth, intensifying production challenges, particularly feed contamination by mycotoxins (Mottet and Tempio, 2017). Corn, which constitutes approximately 65% of poultry diets in the United States, is highly susceptible to fungal growth and mycotoxin formation. These toxic secondary metabolites are commonly detected in crops, feed, and food commodities at both pre‑ and post‑harvest stages (Choudhary and Kumari, 2010).
Recent surveys confirm the widespread presence of mycotoxins in poultry feed. The 2023 dsm‑firmenich survey reported contamination in 88% of U.S. corn and corn by‑products, with 92% of finished poultry diets containing multiple mycotoxins (dsm‑firmenich, 2023). In the Midwestern United States, fumonisins (FUM), deoxynivalenol (DON), zearalenone (ZEN), and aflatoxins (AF) account for over 95% of mycotoxicosis cases (Weaver et al., 2021). Globally, fumonisin, aflatoxin, ochratoxin, DON, and ZEN are the most frequently detected mycotoxins, influenced by climatic conditions such as temperature, humidity, and drought (Greco et al., 2014; Gruber‑Dorninger et al., 2019).
Mycotoxin exposure reduces feed intake and nutrient utilization, increases susceptibility to enteric pathogens, and causes economic losses estimated at USD 0.5–1.5 billion annually (Agboola et al., 2015; Desjardins et al., 1992). Mycotoxin binders (MTB) mitigate these effects by adsorbing toxins in the gastrointestinal tract, are GRAS‑classified by the U.S. FDA, and require in vitro and in vivo validation for efficacy and nutrient safety in the EU (Di Gregorio et al., 2014; Gimeno and Martins, 2007; European Commission, 2006; Barrientos‑Velazquez et al., 2016).
2. Classification of Mycotoxin Binders
Mycotoxin binders are classified by their nature into two major groups: 1) inorganic binders constituted by silicate minerals and activated carbon (AC) binders, and 2) organic binders constituted by yeast cell wall (YCW) or micro-ionized fiber extracted from different plant materials (Figure 1).
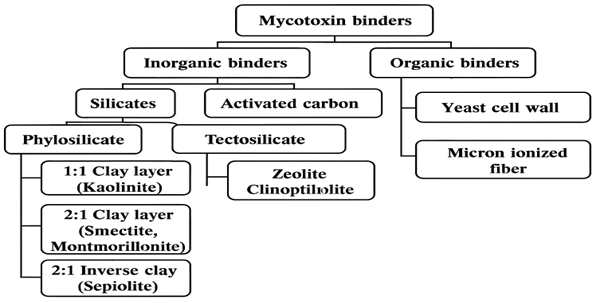
Figure 1. A diagram representing the classification of different mycotoxin binders by their source, nature and structural composition.
2.1. Inorganic binders
There is no consensus on the classification of clay binders that is acceptable to different disciplines such as agriculture, environment, or construction applications. Therefore, we report a classification of inorganic binders based on their properties to bind mycotoxins as proposed and updated by Murray (2007).
a) Silicate binders
Silicates are the most abundant elements found on earth crust (Kandel, 2018). Silicate is a mineral combining silicon dioxide (SiO2 4−) with a tetrahedral structure, where the silicon ion is in the center and surrounded by four oxygen atoms. The interaction of the positive silicon charges and negative oxygen charges results in an unbalanced structure. This allows the free oxygen charges to be bound to other silicon ions forming a chain of tetrahedral structures in different combinations, resulting in chains, sheets, rings, and three-dimensional structures. The tetrahedral sheet is the basis of silicate binders where different subgroups of silicate are formed in combination with other mineral ions in bi or three-dimensional structures. The two main subclasses of silicates are phyllosilicate (sheets of silicate) or tectosilicate (framework silicate, Figure 2).
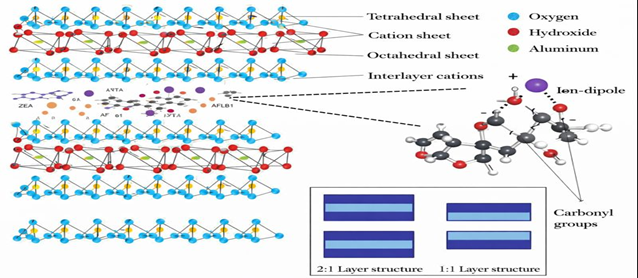
Figure 2. Molecular structure of octahedral and tetrahedral sheets of tectosilicate binders, and an illustration of the contribution of ions to the adsorption mechanism of mycotoxins.
b) Phyllosilicate binders
Phyllosilicates are bidimensional laminar or tubular minerals composed of tetrahedral silicate sheets linked to octahedral sheets of aluminum or magnesium hydroxides [(Al/MgOH)₆]. Charge imbalance within the octahedral layer, compensated by either two Al³⁺ or three Mg²⁺ ions, results in dioctahedral or trioctahedral structures. Based on layer stacking, phyllosilicates are classified as 1:1 types (e.g., kaolinite–serpentinite), consisting of one tetrahedral and one octahedral sheet, and 2:1 types (e.g., smectites), where an octahedral sheet is sandwiched between two tetrahedral sheets.
Isomorphic substitution of Si⁴⁺ or Al³⁺ with lower‑valence cations (e.g., Mg²⁺, Fe²⁺) generates negatively charged layers balanced by exchangeable interlayer cations, conferring swelling behavior and high cation‑exchange capacity essential for mycotoxin adsorption. Smectites, particularly montmorillonite, exhibit high adsorption efficiency, while bentonite, rich in montmorillonite, shows comparable binding properties (Murray, 2007).
c) Tectosilicate binders
Tectosilicate binders are crystalline aluminosilicate minerals, with zeolites as the main constituents. They are formed by three-dimensional assemblies of tetrahedral units linked through shared oxygen atoms, creating cage- or ring-like porous structures. These uniform pores contain exchangeable cations and water molecules, providing adsorption sites where potassium and calcium ions interact with mycotoxins depending on molecular size. Zeolites are classified based on crystal structure, chemical composition, cation type, pore size, and structural stability. Clinoptilolite is the most widely used zeolite due to its high resistance to low pH and elevated temperatures, functioning as a molecular sieve with pore sizes of approximately 3–8 Å. Thermal treatment or cation enrichment can further enhance its adsorption capacity (Eseceli et al., 2017).
d) Activated carbon
Activated carbon (AC) is an insoluble carbonaceous powder produced by pyrolysis of organic materials such as wood, bamboo, or coal at temperatures up to 2000 °C. An activation process is required to enhance its adsorption capacity by developing a highly porous structure. Chemical activation involves impregnation with agents such as KOH, H₃PO₄, or ZnCl₂ followed by heating, but often results in impurities and environmentally harmful residues. Physical activation uses oxidation with oxygen or CO₂ at 600–900 °C, producing highly microporous carbon with a large surface area (500–3000 m²/g). Adsorption efficiency is directly related to pore availability, with AC sources showing variable mycotoxin-binding capacity (Galvano et al., 1997).
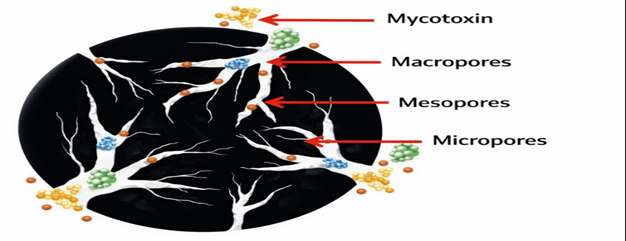
Figure 3. Structure of macro and micropores of activated carbon for the adsoprtion of mycotoxins and other nutrients.
2.2. Organic binders
a) Yeast Cell Wall (YCW)
The yeast cell wall (15–30% of yeast dry weight) is the main component responsible for mycotoxin adsorption. It consists of an inner layer rich in β-(1,3)- and β-(1,6)-D-glucans (50–60%), which provide structural rigidity and binding sites, linked to the membrane by chitin. Excess chitin reduces flexibility and mycotoxin affinity. The outer layer (≈40%) is composed of glucomannans and mannoproteins that determine surface properties. Mycotoxin-binding capacity increases with higher β-D-glucan content in the yeast strain (Jouany et al., 2005).
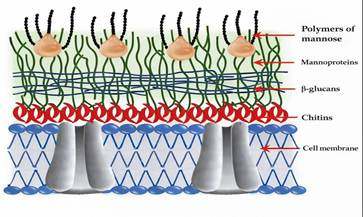
Figure 4. The composition of different yeast cell wall sheets and their components (adapted from Talavera et al., 2013).
b) Micro-ionized fiber
Micro‑ionized fibers are emerging mycotoxin binders (MTB) capable of adsorbing a wide range of mycotoxins. Various plant‑derived biomaterials, including grape pomace and stem, olive pomace, alfalfa hay, and wheat straw, have shown binding efficiencies ranging from 27 to 90%, depending on the material and mycotoxin type. Their adsorption relies on physico‑chemical interactions between mycotoxins and fiber components such as lignin, cellulose, and polyphenols, similar to silicate or activated carbon binders. However, high inclusion rates (≈20 kg/t) limit their use in monogastric diets, while ruminant diets may better tolerate them (Čolović et al., 2019).
3. Adsorption Mechanism of Different Binders
3.1. Mycotoxin binder properties
a) Silicate binders (clays and zeolites)
Silicate binders adsorb mycotoxins mainly through cation exchange capacity (CEC) and surface charge interactions, which are strongly influenced by pH and point of zero charge (PZC). At low pH, protonation reduces adsorption, whereas higher pH exposes negative charges that facilitate binding of cations interacting with mycotoxin carbonyl groups via weak ion‑dipole and Van der Waals force. Interlayer spacing is critical; sodium bentonite shows greater aflatoxin adsorption than calcium bentonite, while zeolites are limited by smaller pore size. Structural and organic modifications further enhance adsorption efficiency (Jaynes & Zartman, 2011).
b) Activated carbon (AC)
Activated carbon adsorbs mycotoxins primarily via hydrophobic interactions and π‑bonding, showing greater affinity for non‑polar toxins. Activation processes increase surface oxygen‑containing functional groups, enhancing polarity and enabling adsorption of polar mycotoxins such as aflatoxins and fumonisins. Adsorption efficiency is determined by surface area and pore size distribution, which must match mycotoxin molecular dimensions (Goto et al., 2015). The adsorption efficiency of activated carbon (AC) is strongly influenced by pore size and pore size distribution.
AC pores are classified into three categories: micropores (<2 nm), mesopores (2–50 nm), and macropores (>50 nm). Micropores contribute most to surface area and adsorption capacity, while meso- and macropores facilitate diffusion. If the pore size is not compatible with the molecular size of mycotoxins, diffusion into the pores is restricted. Limited accessibility to the internal pore surface reduces overall adsorption efficiency of AC.
c) Yeast cell wall (YCW)
Yeast cell wall binders act mainly through β‑ (1,3)‑D‑glucans, which interact with mycotoxins via Van der Waals forces between aromatic rings and glucan structures, as well as hydrogen bonding with hydroxyl, ketone, and lactone groups. Three‑dimensional conformational compatibility between the mycotoxin and glucan helices enhances complex stability and binding strength (Yiannikouris et al., 2004).
4. Mycotoxin properties
Physico-chemical characteristics of mycotoxins significantly influence the adsorption capacity of MTB (Galvano et al., 1997).
Classification of mycotoxins can be based on:
- Polarity
- Solubility
- Chemical structure (Figure 5)
a) Polarity
Polarity reflects the charge distribution within a mycotoxin molecule. Mycotoxins can be classified as polar, non-polar, or intermediate. Aflatoxins (AF) and fumonisins (FUM) are the most polar mycotoxins. Zearalenone (ZEA) is non-polar. Deoxynivalenol (DON), T-2 toxin, and ochratoxin A (OTA) exhibit intermediate polarity.
b) Solubility
Solubility of mycotoxins in the surrounding medium is crucial for effective adsorption. Most mycotoxins are soluble in organic solvents such as methanol, acetonitrile, and acetone. Water solubility depends on polarity: More polar mycotoxins are generally more soluble in water.
c) Chemical structure, size, and shape
These structural features strongly affect the adsorption efficiency of mycotoxins. Aflatoxins (AF) are small, flat molecules, allowing easy entry into the interlayer spaces of binders, leading to higher adsorption. Fumonisins (FUM) have a large, branched molecular structure, which restricts their access to the interlayer space of MTB, resulting in reduced adsorption (Galvano et al., 1996).
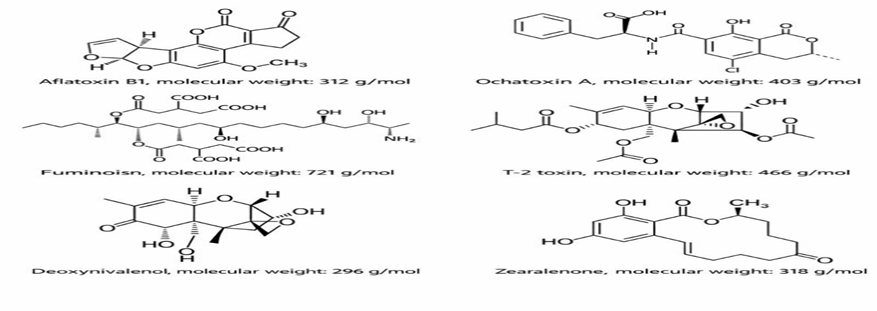
Figure 5. Chemical structure of the major mycotoxins and their molecular weight.
Table 1. Different types of Mycotoxins
| Name | Source | Types / Characteristics | Target organ | Harmful effects |
| Aflatoxin | Aspergillus flavus, Aspergillus parasiticus | Naturally occurring aflatoxins include B1, B2, G1, and G2; among these, B1 is found in the highest concentration and is the most toxic | Liver, Kidney, Spleen, Testes, Thymus | Loss of egg production, anaemia, haemorrhage, liver damage, paralysis, poor FCR, immunosuppression |
| Ochratoxin | Aspergillus ochraceus, Penicillium verridicatum | Four types: A, B, C, and D; Ochratoxin A is the most toxic | Kidney, Liver, Thymus | Gout, immunosuppression, reduced weight gain, reduced egg production and egg weight |
| Trichothecene (T-2 toxin) | Fusarium spp. | — | — | Reduced feed intake, decreased growth, immunosuppression |
| Citrinin | Penicillium spp. | — | Kidney | Wet droppings, reduced weight gain |
| Oosporein | — | — | Kidney | Gout, high mortality |
| Moniliformin | Fusarium moniliforme | — | — | Reduced feed intake and body weight; in chicks causes heart damage and death; in layers reduces rate of laying and delays peak production |
| Fumonisins | Fusarium moniliforme | — | — | More harmful to pigs and horses; poultry are more resistant |
| Zearalenone | — | — | — | — |
| Ergotism | — | — | — | Reduced feed intake and growth; death of tissues of beak, comb, and toes; diarrhoea; vesicles and crusts on comb and wattle; reduced egg production |
| Fusarochromanone | — | — | — | Leg deformities in chicks |
Table 2. Summary of studies that determine the capacity of different mycotoxin binders to adsorb nutrients
| Binder | Nutrient interaction effects | Observation | Reference |
| Bentonite | High adsorption of vitamins E, B1, B2, and B6, and amino acids (lysine, methionine, threonine); low adsorption of vitamins A, D, and B3 | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| High adsorption of vitamin B1 and pepsin; no adsorption of vitamins D and E | In vitro gastric fluid simulation | Barrientos-Velázquez et al. (2016) | |
| High adsorption of vitamins B12 and B8; no adsorption of vitamin B5 | In vitro gastric fluid simulation and real gastric fluid | Vekiru et al. (2007) | |
| High adsorption of vitamin B6; adsorption of Zn and Co; no adsorption of Cu and Mn | In vitro aqueous solution | Tomasevic-Canovic et al. (2000) | |
| Adsorption of vitamin B2 | In vitro aqueous solution | Mortland and Lawless (1983) | |
| No adsorption of vitamin A | In vivo in chicks | Pimpukdee et al. (2004) | |
| No adsorption of vitamin A | In vivo in chicks | Afriyie-Gyawu (2004) | |
| MMT | High adsorption of vitamins E, B1, B2, and B6, and amino acids (lysine, methionine, threonine); low adsorption of vitamins A, D, and B3 | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Adsorption of vitamin B1 | In vitro gastric fluid simulation | Ghanshyam et al. (2009) | |
| Adsorption of protein, urea, and antibiotics | In vitro agar culture | Pinc (1962) | |
| No adsorption of vitamins A, D, E, B1, and B6 | In vivo in dairy cows | Kihal et al. (2022) | |
| No adsorption of vitamins A and B1 | In vivo in dairy cows | Maki et al. (2016) | |
| Ca-MMT | High adsorption of vitamins E, B1, B2, and B6, and amino acids (lysine, methionine, threonine); low adsorption of vitamins A, D, and B3 | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Adsorption of vitamins B8 and B12 | In vitro simulation of gastric fluid and real gastric fluid | Vekiru et al. (2007) | |
| Clinoptilolite | High adsorption of vitamins E, B1, B2, and B6, and amino acids (lysine, methionine, threonine) | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| No adsorption of vitamins A, D, and B3; no adsorption of amino acids (tryptophan, phenylalanine) | In vitro aqueous solution | Tomasevic-Canovic et al. (2000) | |
| HSCAS | No adsorption of vitamins A, B1, and minerals (Zn, Mn) | In vivo in chicks | Chung et al. (1990) |
| Sepiolite | High adsorption of vitamins E, B1, B2, and B6, and amino acids (lysine, methionine, threonine); low adsorption of vitamins A, D, and B3 | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
| Zeolite | High adsorption of vitamins E, B1, B2, and B6, and amino acids (lysine, methionine, threonine); low adsorption of vitamins A, D, and B3 | In vitro simulation of gastrointestinal tract | Kihal et al. (2020, 2021) |
¹MMT, montmorillonite.
²AC, activated carbon.
³HSCAS, hydrated sodium calcium aluminosilicate.
Table 3. Regulatory guidance levels for major mycotoxins in finished poultry feed as established by the European Union (EU) and United States Food and Drug Administration (FDA). Values are expressed in mg/kg diet. EU limits in finished feed set according to the European Commission Recommendation 2006/576/EC and the European Commission Directive 2003/100/EC; USA limits in finished feed set according to the Food and Drug Administration regulatory guidance for toxins and contaminants.
| Guidance | Deoxynivalenol (DON) (mg/kg) | Fumonisins (FUM) (mg/kg) | Zearalenone (ZEA) (mg/kg) | Aflatoxin (AF) (mg/kg) | Ochratoxin A (OTA) (mg/kg) | T-2 Toxin (mg/kg) |
| EU limits (EC guidance) ¹ | 5 | 20 | – | 0.02 | 0.1 | No guidance level established |
| USA limits (FDA guidance) ² | 5 | 50 | No guidance level established | 0.1 | No guidance level established | No guidance level established |
Table 4. Ranges of ratios of mycotoxin binder to mycotoxins doses used in in vitro tests to determine the mycotoxin adsorption capacity of different mycotoxin binders
| Binders | AFB1 (mg: µg) | DON (mg: µg) | FUM (mg: µg) | OTA (mg: µg) | T-2 (mg: µg) | ZEA (mg: µg) |
| AC | 1:0.008–1:461 | 1:0.2–1:90 | 1:0.92–1:25 | 1:0.025–1:125 | 1:0.1 | 1:0.05–1:20 |
| Bentonite | 1:0.0007–1:200 | 1:0.2–1:12 | 1:2–1:20 | 1:0.002–1:12.5 | 1:0.1–1:0.2 | 1:0.1–1:20 |
| Clinoptilolite | 1:0.2–1:40 | 1:1.2 | – | 1:2 | – | 1:0.05–1:12 |
| HSCAS | 1:0.002–1:600 | 1:0.4–1:12 | 1:2–1:20 | 1:0.025–1:10 | 1:0.1 | 1:0.001–1:20 |
| MMT | 1:0.002–1:20 | 1:0.5–1:12 | 1:2.5 | 1:0.025 | 1:0.1 | 1:0.05–1 |
| Sepiolite | 1:1.6–1:10 | 1:2–1:12 | 1:2 | 1:10 | – | 1:0.5 |
| YCW | 1:0.001–1:461 | 1:0.2–1:12 | 1:2–1:20 | 1:0.001–1:10 | 1:0.1 | 1:0.001–1:20 |
| Zeolite | 1:0.002–1:20 | 1:0.2–1:10 | 1:0.2–1:20 | 1:0.016–1:5 | 1:0.2 | 1:0.05–1:20 |
1AC, activated carbon; HSCAS, hydrated sodium calcium aluminosilicate; MMT, montmorillonite; YCW, yeast cell wall. 2AFB1, aflatoxin B1; DON, deoxynivalenol; FUM, Fumonisin; OTA, ochratoxin; T-2, T-2 toxin; ZEA, zearalenone.
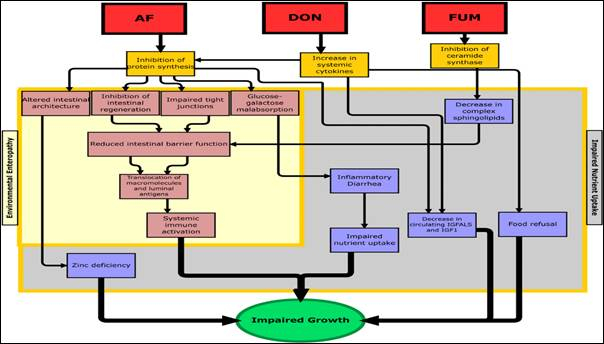
Figure 6. A conceptual framework for the effect of mycotoxin exposure on growth retardation (Smith et.al.2012).
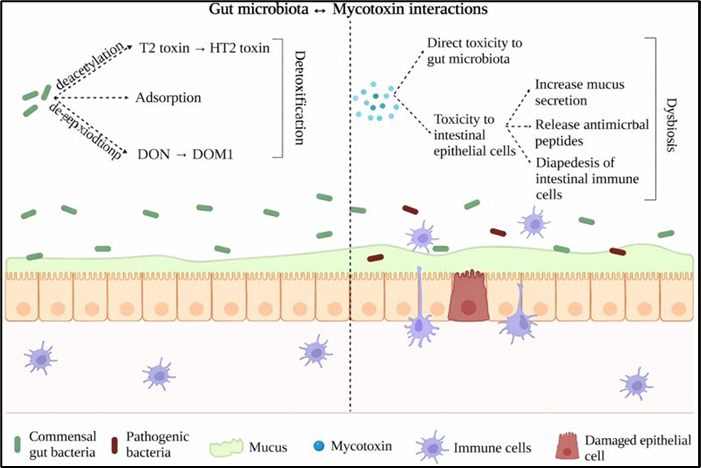
Fig. 7. Gut microbiota and mycotoxins interactions. Illustration of the bidirectional interactions between gut microbiota and dietary mycotoxins in poultry. Mycotoxins can disrupt microbial community structure, reduce beneficial populations, and impair microbially derived functions, including short-chain fatty acid (SCFA) production and mucosal barrier maintenance. Conversely, the gut microbiota can biotransform certain mycotoxins into less toxic metabolites or modulate host responses to exposure. Disruption of this balance may compromise gut integrity, immunity, and overall performance.
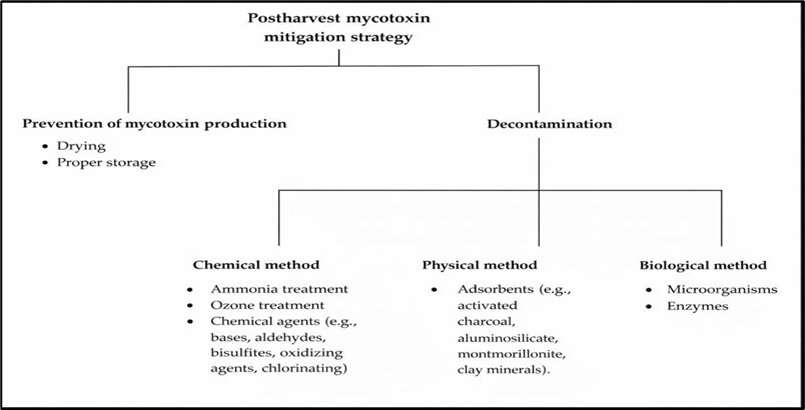
Figure 8. Diagrammatic representation for postharvest mycotoxin mitigation strategies in broiler and layer chickens’ production.
Stallen offers a comprehensive range of world‑class mycotoxin binders, including D‑Tox and Alusil MOS Plus, both of which provide broad‑spectrum protection against diverse mycotoxins commonly found in poultry feed.
5.2. The characteristic features of D-Tox compare with other common toxin binder.
- Binding efficacy of D-Tox to various mycotoxins
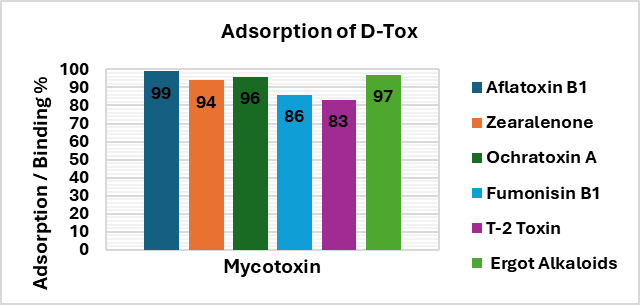
- Features of D-TOX

5.3. D-Tox Benefits:
D‑Tox provides effective and comprehensive control of all major categories of mycotoxins, thereby improving performance and productivity in poultry.
6. Alusil MOS Plus
Alusil MOS Plus contains HSCAS (Activated Hydrated Sodium Calcium Aluminosilicates), activated charcoal, MOS (Mannan Oligosaccharides), copper oxinate, organic acids (propionic, benzoic, acetic, and sorbic acids), lipotropic agents, and spirulina.
6.1. Mechanism of action of Alusil MOS Plus
HSCAS acts as an enterosorbent that tightly and selectively binds aflatoxins in the GI tract of animals decreasing their bioavailability and associated toxicity. Mannan Oligosaccharides act as bio-binder which helps absorption of pathogens, improves intestinal function & immune modulation. Organic acids kill harmful bacteria and fungi. Activated charcoal which is 200 MT grade helps to bind pesticides and toxins like ochratoxin. Copper oxinate is broad spectrum anti-fungal agent which acts against spp. of Aspergillus, Fusarium, Penicillium, Candida etc. Lipotropic agent and herbal ingredients help in mobilizing fat which are accumulated in liver due to damage caused by toxins. Spirulina helps in restoring the liver damaged by toxins.
6.2. The characteristic features of Alusil MOS Plus over other toxin binder.
| Component | Function |
| HSCAS | Enters bloodstream and binds to aflatoxins. |
| Activated Charcoal | Binds multiple mycotoxins including aflatoxins, ochratoxins A, zearalenone, and along with bacterial endotoxins & pesticides in the intestinal tract, preventing their reabsorption. |
| MOS | Binds to pathogenic bacterial fimbriae, increases mucin secretion & acts as a bactericidal agent. |
| Copper Oxinate | Acts as antifungal agent by inhibiting mould growth. |
| Organic Acids | Acetic, benzoic, propionic & sorbic acids inhibit fungal growth and reduce aflatoxin secretion. |
| Lipotropic Agent | Aids in mobilising fat accumulated in liver due to toxin damage. |
| Spirulina | Helps restore liver function damaged by toxins. |
6.3. Alusil MOS Plus Benefits:
Alusil MOS Plus acts as a broad-spectrum mould inhibitor and supports bio-neutralization of mycotoxins, helping prevent toxin-related damage and overcome clinical symptoms of aflatoxicosis. It protects the immune system, enhances vaccine and drug response, improves pellet quality, functions as an anti-caking agent in feed, and does not bind essential vitamins and minerals.
7. Conclusion
Stallen South Asia Pvt. Ltd. offers effective toxin binders, D-tox and Alusil MOS Plus, which are helpful in the detoxification of mycotoxins in poultry. D-tox provides broad‑spectrum control of both polar and non‑polar mycotoxins through pH‑stable adsorption and pre‑absorptive detoxification, without binding amino acids, fat‑soluble vitamins, minerals, or micronutrients. It also sequesters heavy metals, endotoxins, and biogenic amines, reducing immunosuppression. Alusil MOS Plus alleviates aflatoxicosis, protects immune function, enhances vaccine and drug response, and improves feed quality through anticaking and pellet‑stabilizing effects.
References
Agboola, A.F., Omidiwura, B.R.O., Odu, O., Odupitan, F.T., Iyayi, E.A., 2015. Effect of probiotic and toxin binder on performance, intestinal microbiota and gut morphology in broiler chickens. Journal of Animal Science Advances 5(7), 1369. https://doi.org/10.5455/jasa.20150709085312.
Barrientos-Velazquez, A.L., Arteaga, S., Dixon, J.B., Deng, Y., 2016. The effect of pH, pepsin, exchange cation, and vitamins on aflatoxin adsorption on smectite in simulated gastric fluids. Applied Clay Science 120, 17–23. https://doi.org/10.1016/j.clay.2015.11.014.
Čolović, R., Puvača, N., Cheli, F., Avantaggiato, G., Greco, D., Đuragić, O., Pinotti, L., 2019. Decontamination of mycotoxin-contaminated feedstuffs and compound feed. Toxins 11, 617. https://doi.org/10.3390/toxins11110617.
Choudhary, A.K., Kumari, P., 2010. Management of mycotoxin contamination in preharvest and post-harvest crops: present status and future prospects. Journal of Phytology 2.
Desjardins, A., Plattner, R., Shackelford, D., Leslie, J., Nelson, P., 1992. Heritability of fumonisin B1 production in Gibberella fujikuroi mating population A. Applied and Environmental Microbiology 58, 2799–2805.
Di Gregorio, M.C., Neef, D.V., Jager, A.V., Corassin, C.H., Carao, A.C.P., Albuquerque, R., Azevedo, A.C., Oliveira, C.A.F., 2014. Mineral adsorbents for prevention of mycotoxins in animal feeds. Toxin Reviews 33, 125–135. https://doi.org/10.3109/15569543.2014.905604.
Eseceli, H., Ozcan, M.A., Demir, E., Bilal, T., 2017. Increasing the adsorption of clinoptilolite as toxin binder by heat treatment. Bulgarian Journal of Veterinary Medicine 20, 18–26.
European Commission (EC), 2006. EC regulation No 401/2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. Official Journal of the European Communities L 70, 12–34.
Food and Drug Administration (FDA), 2001. Guidance for Industry: Fumonisin levels in human foods and animal feeds; Final guidance. US FDA, Silver Spring, MD, USA.
Galvano, F., Pietri, A., Bertuzzi, T., Bognanno, M., Chies, L., Angelis, A., Galvano, M., 1997. Activated carbons: in vitro affinity for fumonisin B1 and relation of adsorption ability to physicochemical parameters. Journal of Food Protection 60, 985–991. https://doi.org/10.4315/0362-028X-60.8.985.
Gimeno, A., Martins, M.L., 2007. Micotoxinas y micotoxicosis en animales y humanos. 1st ed. Miami, FL, USA: Special Nutrients; p. 128.
Goto, T., Amano, Y., Machida, M., Imazeki, F., 2015. Effect of polarity of activated carbon surface, solvent and adsorbate on adsorption of aromatic compounds from liquid phase. Chemical and Pharmaceutical Bulletin 63, 726–730. https://doi.org/10.1248/cpb.c15-00039.
Greco, M.V., Franchi, M.L., Rico Golba, S.L., Pardo, A.G., Pose, G.N., 2014. Mycotoxins and mycotoxigenic fungi in poultry feed for food-producing animals. The Scientific World Journal, 1–10. https://doi.org/10.1155/2014/968215.
Gruber-Dorninger, C., Jenkins, T., Schatzmayr, G., 2019. Global mycotoxin occurrence in feed: a ten-year survey. Toxins 11, 375. https://doi.org/10.3390/toxins11070375.
Jaynes, W.F., Zartman, R.E., 2011. Aflatoxin toxicity reduction in feed by enhanced binding to surface-modified clay additives. Toxins 3, 551–565. https://doi.org/10.3390/toxins3060551.
Jouany, J.P., Yiannikouris, A., Bertin, G., 2005. How yeast cell wall components can alleviate mycotoxicosis in animal production and improve the safety of edible animal products. Journal of Animal Science 14, 171–190. https://doi.org/10.22358/jafs/70361/2005.
Kandel, R., 2018. Potential for using aluminosilicates for removal of heavy metals and mycotoxins from feed and water [Master’s thesis]. Ås, Norway: Norwegian University of Life Sciences.
Mottet, A., Tempio, G., 2017. Global poultry production: current state and future outlook and challenges. World’s Poultry Science Journal 73, 245–256.
Murray, H.H., 2007. Developments in clay science: Occurrences, processing and applications of kaolins, bentonites, palygorskite–sepiolite, and common clays, Vol. 2. UK: Elsevier Science & Technology; pp. 1–180.
Smith, L., Stoltzfus, R., Prendergast, A., 2012. Food chain mycotoxin exposure, gut health, and impaired growth: A conceptual framework. Advances in Nutrition 3, 526–531. https://doi.org/10.3945/an.112.002188.
Weaver, A.C., Weaver, D.M., Adams, N., Yiannikouris, A., 2021. Co-occurrence of 35 mycotoxins: a seven-year survey of corn grain and corn silage in the United States. Toxins 13, 516.
Yiannikouris, A., Francois, J., Poughon, L., Dussap, C.G., Bertin, G., Jeminet, G., Jouany, J.P., 2004. Adsorption of zearalenone by β-D-glucans in the Saccharomyces cerevisiae cell wall. Journal of Food Protection 67, 1195–1200. https://doi.org/10.4315/0362-028X-67.6.1195.






