Dr. Amit V. Janbandhu & Dr. Sanjay Singhal
1. Introduction
The poultry industry is among the most efficient sectors of agriculture, making a significant contribution to livelihood generation and global nutritional security. The global poultry population currently exceeds 26.8 billion birds (FAO, 2020). From 1961 to 2019, worldwide poultry meat production increased to approximately 132 million tonnes annually, representing nearly 37% of total global meat production (FAO, 2020). This growing demand for animal‑derived foods is primarily driven by rapid population growth, rising income levels, and increasing urbanization (FAO, 2020).
To satisfy the rising demand for meat and eggs, modern poultry production systems operate under intensive rearing conditions, exposing birds to continuous physiological stress. As a result, antibiotics have been extensively used for disease prevention, growth promotion, and immune enhancement. However, the indiscriminate use of antibiotics has hastened the emergence of antimicrobial resistance among pathogenic bacteria (Garcia‑Migura et al., 2014; Roth et al., 2019). The World Health Organization has identified antimicrobial resistance as “a serious threat to public health worldwide that requires action across all government sectors and society” (WHO Factsheets, 2015). Antibiotic‑resistant bacteria can enter the human food chain through animal‑derived products, posing substantial public health risks (Cui et al., 2005). Moreover, fresh meat products may act as reservoirs of antibiotic‑resistance genes that can be transferred to humans through regular consumption (Diarrassouba et al., 2007). In response to the escalating challenge of antimicrobial resistance, the livestock industry is increasingly exploring effective alternatives to conventional antibiotics, with probiotics emerging as a promising and sustainable solution.
2. What Is Probiotics?
The concept of probiotics was first introduced in the early 1900s by the Russian‑born Nobel laureate Elie Metchnikoff, who demonstrated that the regular consumption of beneficial microorganisms could positively influence gastrointestinal health (Metchnikoff, 1907). His observations of populations that consumed fermented milk products led to the hypothesis that beneficial intestinal microflora enhance resistance to pathogenic organisms. The term probiotics is derived from Greek, meaning “pro‑life” (Shokryazdan et al., 2017a, 2017b). According to the FAO/WHO, probiotics are defined as “live organisms which, when administered in adequate amounts, confer a health benefit on the host” (FAO/WHO Joint Report, 2001).
Probiotics are widely recognized for their ability to modulate gut microflora and enhance immune responses (Chen et al., 2012) and are extensively applied in both clinical and veterinary practices (Abushelaibi et al.). In livestock production, probiotic supplementation has been associated with improved growth performance, production efficiency, disease resistance, nutrient digestibility, immune function, and fecal microbial balance (Lan et al., 2017). In recent years, non‑specific immunomodulators—including probiotics, prebiotics, synbiotics, postbiotics, polysaccharides, organic acids, enzymes, and essential oils—have gained considerable attention as alternatives to conventional antibiotics and are increasingly used to promote gut health in poultry birds (Callaway et al., 2017).
3. Probiotic and Related Biotic Agents in Poultry
The species currently being used in probiotic preparations are varied and many. These are mostly Lactobacillus bulgaricus, Lactobacillus acidophilus, Lactobacillus casei, Lactobacillus helveticus, Lactobacillus lactis, Lactobacillus salivarius, Lactobacillus plantarum, Streptococcus thermophilus, Enterococcus faecium, Enterococcus faecalis, Bifidobacterium spp. and Escherichia coli. With two exceptions, these are all intestinal strains. The two exceptions, Lactobacillus bulgaricus and Streptococcus thermophilus, are yoghurt starter organisms (Fuller et.al, 1989). Some other probiotics are microscopic fungi such as strains of yeasts belonging to Saccharomyces cerevisiae species (Guillot et.al, 1998).
In broiler nutrition, probiotic species belonging to Lactobacillus, Streptococcus, Bacillus,
Bifidobacterium, Enterococcus, Aspergillus, Candida, and Saccharomyces have a beneficial effect on broiler performance (Tortuero et.al, 1973), modulation of intestinal microflora and pathogen inhibition, intestinal histological changes, immunomodulation, certain haematobiochemical parameters, improving sensory characteristics of dressed broiler meat and promoting microbiological meat quality of broilers (Kabir et.al, 2005).
The International Scientific Association of Probiotics and Prebiotics defines prebiotics as “a substrate that is selectively utilized by host microorganism conferring a health benefit” (Gibson et al., 2017). Synbiotics are defined as a “synergistic combination of probiotics and prebiotics that are beneficial for the host by improving the development and colonization of live microorganisms in the gut” (FAO/WHO joint report, 2002). Postbiotics are defined as “the preparation of inanimate microorganism and/or their components that confer a health benefit on the host” (Salminen et al., 2021).
Given their safety, efficacy, and sustainability, probiotics have gained considerable attention as viable alternatives to antibiotics. This article outlines the status, mechanisms, and use of probiotics in poultry as alternatives to antibiotics.”
4. The concept of probiotics
In healthy, non-stressed poultry, a dynamic balance exists between beneficial and non-beneficial gut bacteria, which is essential for optimal performance. Stress disrupts this balance by reducing beneficial flora, particularly lactobacilli, allowing overgrowth of harmful microorganisms. This imbalance may result in clinical conditions such as diarrhea or subclinical effects that impair growth and feed efficiency. Although the protective gut microflora is relatively stable, it can be influenced by key factors including excessive hygiene, antibiotic use, and stress. Under natural conditions, chicks acquire a complete and protective gut microflora through contact with the hen, providing resistance against infection. In contrast, commercially reared chicks hatch in sanitized incubators lacking normal intestinal microorganisms. Gut colonization may be influenced by eggshell microbiota and the onset of gastric HCl secretion at approximately 18 days of incubation, which affects microbial selection. Consequently, early probiotic supplementation is particularly important in poultry, as chicks are deprived of maternal microbial transfer and can benefit from microbial preparations that restore protective gut microflora (Fuller et al., 2001).
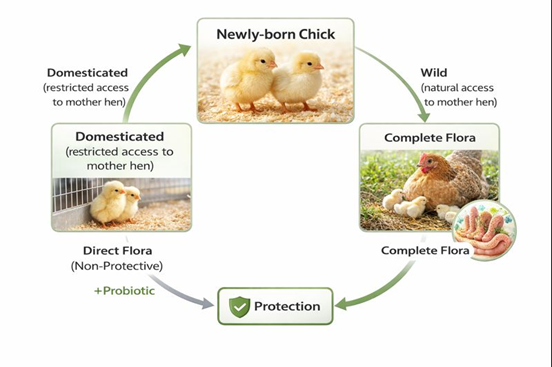
Figure 1. Schematic representation of the concept of probiotics (modified from (Fuller et.al, 2001).
5.Mechanism of action of probiotics
Probiotics place a key role in gut microbial health. The mechanisms of action of probiotics mainly include two, i.e. competitive exclusion and immune system modulation.
Competitive exclusion of pathogens by probiotics includes:
(a) production of inhibitory compounds like bacteriocins, mucins, defensins, etc. (b) preventing the adhesion of pathogens, (c) competition for nutrients, (d) reduction of toxin bioavailability, and (e) modulation of the host immune system including the enhancement of both innate and adaptive immunity (Hernandez-Patlan et al., 2020).
a) Secretion of inhibitory compounds
Probiotics inhibit pathogenic bacteria through the production of antimicrobial compounds, including antimicrobial peptides (AMPs) such as bacteriocins, as well as organic acids, hydrogen peroxide, ethanol, diacetyl, and carbon dioxide (Liao & Nyachoti, 2017). Bacteriocins are ribosomally synthesized AMPs that suppress pathogens by disrupting cell wall synthesis or forming membrane pores while sparing beneficial gut microbiota (Cotter et al., 2013). Pediocin A from Pediococcus pentosaceus, divercin from Carnobacterium divergens, and nisin from Lactococcus lactis have shown inhibitory effects against Clostridium perfringens and improved broiler performance (Grilli et al., 2009). Synergistic bacteriocin activity with other biomolecules has been reported against multiple pathogens (Rishi et al., 2014). Organic acids reduce intracellular pH and disrupt bacterial metabolism and membranes, while lactic acid bacteria inhibit Salmonella, Listeria monocytogenes, and Escherichia coli without harming intestinal epithelium (Ricke, 2003). Additional inhibitory effects are mediated by ethanol, diacetyl, and carbon dioxide (Ingram, 1989).
b) Inhibition of pathogenic adhesion
Probiotics prevent pathogen colonization by competitively blocking adhesion sites on intestinal epithelial cells, a key criterion for selecting effective probiotic strains . Probiotic adhesion stimulates mucosal immunity and promotes the secretion of mucins and defensins, thereby strengthening the epithelial barrier (Bermudez-Brito et al., 2012). Mucins are highly glycosylated glycoproteins that form the mucus layer and inhibit pathogen attachment and colonization (Collado et al., 2005). Interactions between probiotic surface proteins and intestinal epithelial cells further exclude pathogens. Defensins, small cationic antimicrobial peptides, inhibit bacterial growth by disrupting membranes or cell wall synthesis and can neutralize bacterial toxins (Ayabe et al., 2000).
c) Competition for nutrients
Probiotics limit pathogen growth by competing for essential nutrients and occupying intestinal epithelial adhesion sites, thereby restricting pathogen attachment in the gastrointestinal tract . This competitive exclusion reduces pathogen proliferation and colonization and creates unfavorable conditions for pathogen survival (Callaway et al., 2008). Competitive exclusion has been demonstrated in vitro using chicken intestinal mucosa (Hirn et al., 1992). In vivo studies show that early supplementation with lactobacillus-based probiotics (1 × 10⁵ CFU/mL, 1–7 days of age) significantly reduced Salmonella colonization in chicks (Penha Filho et al., 2015).
d) Reduction in toxin bioavailability
Probiotics like lactobacillus help the reduction in the uptake of pathogenic toxins in the intestinal cells. The positive effects of LAB-based probiotics had helped in the reduction of toxin expression in the gut. Lactic acid bacteria are known for their natural barriers against mycotoxins which are harmful compounds for animals. A few strains can also eradicate the detrimental reactions of aflatoxins on human and animal health (Abbes et al., 2016).
e) Modulation of the host immune system
Probiotics modulate host immunity by interacting with intestinal epithelial cells, dendritic cells, macrophages, and lymphocytes. These interactions enhance innate immune defenses by limiting pathogen proliferation and reinforcing epithelial barriers through increased mucus and antimicrobial peptide production. Intestinal epithelial and dendritic cells recognize probiotics via pattern recognition receptors, initiating immune signaling cascades. Activation of antigen-presenting cells stimulates adaptive immunity through T- and B-cell responses. Probiotics regulate cytokine expression and suppress intestinal inflammation by downregulating TLR and NF-κB signaling pathways. Enhanced IgA and IgG responses have been observed in broilers supplemented with probiotic strains such as Clostridium butyricum and Lactobacillus plantarum (Han et al., 2018).
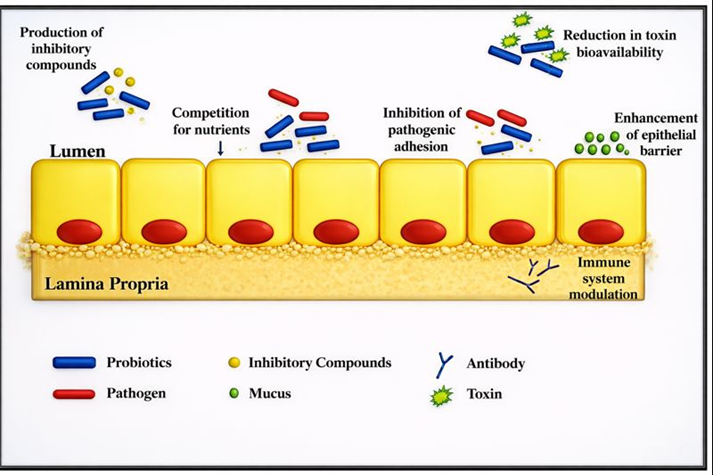
Fig. 2. Mode of action of probiotics. It starts with the secretion of inhibitory compounds leading to inhibition of the pathogen adhesion to the epithelial layer of the GI tract besides creating competition for nutrients among pathogens thereby reducing their colonization. Also, it helps in diminishing the toxin bioavailability and modulates the immune system of the host by activating adaptive and innate immunity.
6. Single- and multi-strain probiotics
Probiotics are classified as single- or multi-strain formulations. Single-strain probiotics contain one microbial species, commonly Lactobacillus, Bifidobacterium, Streptococcus, Pediococcus, Enterococcus, Bacillus, Saccharomyces, and Micrococcus. Multi-strain probiotics combine multiple strains or genera to provide complementary benefits and have been shown to improve growth performance and gut health in broilers, including under disease challenge conditions. Commercial products such as Probios from Stallen South Asia private Ltd contain diverse probiotic combinations. Probiotic efficacy depends on strain composition and viable counts, with variable outcomes reported (Aalaei et al., 2018).
a) Bacillus
Many strains of Bacillus have potential against pathogenic bacteria. A group of researchers isolated 200 Bacillus strains from the faeces of broiler chicken and many strains among them showed activity against C. perfringens in in vitro conditions. A study suggested that B. subtilis strain SP6 when used in a field trial, the mortality of chicken infected with Necrotic enteritis was reduced to half. It also reduced the number of C. perfringens and enhanced the intestinal health of chickens. Regular use of B. licheniformis supplementation reduced mortality and increased the performance among the chicks (Knap et al., 2010).
b) Yeast
Yeasts possess antimicrobial and immunomodulatory properties, largely due to β-glucans that stimulate host immunity. They inhibit pathogens by producing mycocins, degrading toxins, preventing epithelial adhesion, and competing for nutrients. Saccharomyces boulardii improves intestinal health and reduces Salmonella enteritidis infection, while recombinant Pichia pastoris expressing Clostridium perfringens α-toxin enhances broiler performance (Gil de Lossantos et al., 2005).
c) Enterococci
Enterococci produce bacteriocins (enterocins) with activity against Gram-positive and Gram-negative bacteria . Enterococcus faecium supplementation reduces Clostridium perfringens, alleviates coccidiosis, and improves growth performance and nutrient utilization in broilers. Enhanced IgA production, immune responses, and microbiome modulation have also been reported with E. faecium and E. faecalis supplementation (Beirão et al., 2018)
7. Beneficial effects of probiotics on poultry
a) Effects on growth performance and productivity
Probiotics improve body weight gain, feed intake, feed conversion ratio, and overall productivity in poultry. Supplementation with Pediococcus acidilactici and Bacillus subtilis enhances egg quality, increases eggshell thickness, and reduces yolk cholesterol. Multi-strain probiotics improve egg production and mitigate heat-stress effects. Probiotics also enhance meat microbiological quality, reduce Salmonella enteritidis contamination, and improve nutrient metabolism and growth performance (Bailey et al., 2000).
b) Effects on serum biochemistry
Probiotic supplementation significantly modulates serum biochemistry in poultry by reducing total cholesterol, LDL, VLDL, triglycerides, uric acid, and liver enzymes (ALT, AST), while increasing protein and calcium levels . Lactobacillus spp., Enterococcus faecium, and Bacillus subtilis reduce cholesterol absorption and improve lipid metabolism in broilers and layers (Kalavathy et al., 2003).
c) Effect on health and immunity
Probiotics enhance poultry health and immunity by modulating gut microbiota and immune signaling. Lactic acid bacteria (LAB) regulate pro- and anti-inflammatory cytokines (IL-1β, IL-6, IL-10, IFN-γ, TNF-α) and suppress inflammation through NF-κB and TLR-mediated pathways. Supplementation with Clostridium butyricum, Lactobacillus spp., and Saccharomyces cerevisiae enhances gut flora, T-cell responses, intraepithelial lymphocyte activity, and mucosal immunity in broilers (Yang et al., 2012).
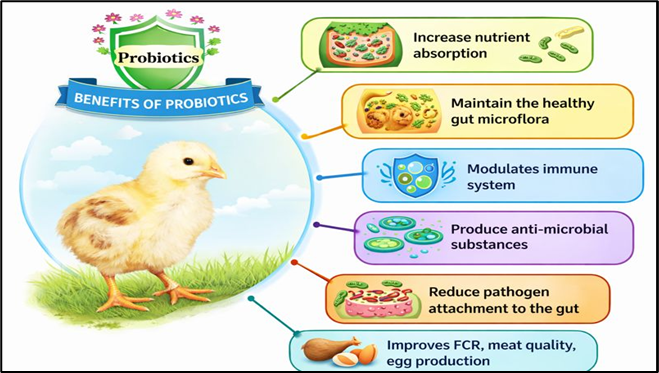
Fig. 3. Positive effects of probiotics on poultry
Table 1. Few studies showing the potentials of probiotics in poultry.
| S. No. | Probiotic species / formulation | Key findings | Reference |
| 1 | Lactobacillus spp. | Reduced Clostridium perfringens load in chicken gut | Gerard et al., 2008 |
| 2 | Lactobacillus spp. | Reduced E. coli and improved gut health | Grilli et al., 2009 |
| 3 | Lactobacillus spp. | Improved growth performance and intestinal morphology | De Cesare et al., 2020 |
| 4 | Pediococcus acidilactici | Improved egg quality and reduced yolk cholesterol | Mikulski et al., 2012 |
| 5 | Bacillus subtilis | Improved egg quality and reduced heat-stress effects | Sobczak & Kozłowski, 2015; Abdelqader, 2020 |
| 6 | Lactobacillus salivarius | Improved BWG and FCR in broilers | Shokryazdan et al., 2017a, 2017b |
| 7 | Lactobacillus plantarum | Improved gut integrity and immune response | Wang et al., 2018 |
| 8 | L. plantarum + P. polymyxa | Reduced ALT, AST, LDL, and uric acid | Wu et al., 2019 |
| 9 | Lactobacillus spp. | Reduced serum cholesterol and triglycerides | Fathi, 2013 |
| 10 | L. sporogenes | Improved serum protein and reduced lipid profile | Arun et al., 2007 |
| 11 | Lactobacillus acidophilus | Improved gut health and metabolic functions | De Cesare et al., 2020 |
| 12 | Enterococcus faecium | Reduced cholesterol and improved immunity | Capcarova et al., 2010 |
| 13 | Lactobacillus salivarius | Reduced cholesterol and LDL | Shokryazdan et al., 2017a, 2017b |
| 14 | Multi-strain probiotics | Improved gut health and performance | Taherpour et al., 2009 |
| 15 | Lactobacillus spp. | Improved nutrient metabolism and absorption | Burgain et al., 2014 |
| 16 | Lactobacillus spp. | Reduced inflammatory cytokines | Chen et al., 2012; Park et al., 2014 |
| 17 | Clostridium butyricum | Enhanced gut flora and immune response | Yang et al., 2012 |
| 18 | Lactobacillus fermentum | Improved T-cell response | Bai et al., 2013 |
| 19 | L. plantarum + L. johnsonii | Suppressed NF-κB activation | Joo et al., 2011; Li et al., 2015 |
| 20 | Lactobacillus spp. | Reduced IL-1β and enhanced TLR4 expression | Li et al., 2015 |
| 21 | Lactococcus lactis | Induced pro-inflammatory immune signaling | Slawinska et al., 2021 |
| 22 | Lactobacillus casei | Increased IELs and mucosal immunity | Tian et al., 2021 |
| 23 | Multi-strain probiotics | Improved growth and gut microbiota | Lambo et al., 2021 |
| 24 | Bacillus subtilis | Reduced C. perfringens and necrotic enteritis | Jayaraman et al., 2013 |
| 25 | Bifidobacterium spp. | Enhanced immune response | Knapp et al., 2010 |
| 26 | Yeast (Saccharomyces cerevisiae) | Improved gut health and immunity | Novak & Vetvicka, 2008 |
| 27 | Saccharomyces boulardii | Improved intestinal health | Rajput et al., 2013 |
| 28 | Enterococcus faecium | Reduced C. perfringens and improved immunity | Cao et al., 2013 |
| 29 | Enterococcus faecium | In vitro anti-C. perfringens activity | Shin et al., 2008 |
| 30 | Enterococcus faecium | Reduced coccidiosis severity | El-Sawah et al., 2020 |
| 31 | Enterococcus faecium | Improved nutrient utilization and metabolism | Zheng et al., 2016 |
Table 2. Impact of probiotics on chicken production
| Probiotic strain | Host age (condition) | Chicken type | Dosage | Duration | Results | Reference |
| Lactobacillus rhamnosus | 70 weeks | Layers | 1 × 10⁹ CFU | 88 weeks | ↑ Egg production | Huang et al., 2023 |
| Bacillus subtilis | Day-old chicks | Broiler | 500 mg/kg | 42 weeks | ↓ mortality, ↑ ADFI and BW, → FCR and ADG | Qiu et al., 2021 |
| Bacillus amyloliquefaciens | Day-old chicks | Broiler | 10⁶ CFU/g | 42 days | ↑ Body weight and average daily weight gain | Mazanko et al., 2022 |
| Pediococcus acidilactici + Lactobacillus plantarum | Day-old chicks | Broiler | 2 × 10⁹ CFU/chick | 42 days | ↑ Feed conversion and production efficiency, ↓ myopathies | Paz et al., 2019 |
| Lactobacillus acidophilus, L. plantarum + Bifidobacterium spp. | Day-old chicks | Broiler | 1.2 × 10⁹ CFU unit/mL | 21 days | ↑ BW, FCR, feed consumption (starter), ↑ breast muscles, liver, heart, kidneys, lungs | Agustono et al., 2022 |
| Lactobacillus casei, L. acidophilus + Bifidobacterium | Day-old chicks | Broiler | 10 mL probiotics/L water (oral) | 42 days | ↑ Eviscerated yield and breast yield, ↓ ADFI, FCR, abdominal fat | Zhang et al., 2021 |
| Bacillus subtilis | Day-old chicks | Broiler | 0.1% probiotics | 42 days | ↑ BW and ADWG | Popov et al., 2024 |
| Bacillus subtilis | Day-old chicks | Broiler | 1.0 × 10⁶ spores/g feed | 42 days | ↑ Bone mineralization, density, size, wall thickness, and tibia/femur weight | Yan et al., 2018 |
| Bifidobacterium spp. (in ovo) | Day-old chicks | Broiler | 2 × 10⁸ CFU | 28 days | ↑ LBW and DBWG | Abdel-Moneim et al., 2020 |
| Lactobacillus fermentum + Saccharomyces cerevisiae | Day-old chicks | Broiler | 1 × 10⁷ CFU/g and 2 × 10⁶ CFU/g | 42 days | ↑ ADG and feed efficiency | Bai et al., 2013 |
| Lactobacillus johnsonii | Day-old chicks | Broiler | 1 × 10⁵ CFU/g BS15/g | 42 days | No changes | Wang et al., 2018a |
| Lactobacillus johnsonii | Day-old chicks | Broiler | 1 × 10⁶ CFU/g BS15 | 28 days | ↑ Starter, finisher, and overall daily weight gain | Wang et al., 2018a |
| Bacillus subtilis | 25 weeks | Layers | 9.0 × 10⁵ CFU/g | 122 days | ↑ Hatchability, fertility, egg weight, yolk colour/index, eggshell thickness | Liu et al., 2019 |
| Bacillus subtilis | 28 weeks | Layers | 1.0 × 10⁵, 1.0 × 10⁶, 1.0 × 10⁷, 1.0 × 10⁸ (B4) CFU/g | 24 weeks | No significant effect on egg production | Guo et al., 2017 |
| Bacillus subtilis | 32 weeks | Layers | 4 × 10⁹ CFU/g | 90 days | No significant effect on ABWG and FCR | Fathi et al., 2018 |
8.Probios
Probios contains nine different species of beneficial microflora, each at a concentration of 2 × 10⁸ CFU. These include Bifidobacterium bifidum, Lactobacillus acidophilus, Lactobacillus bulgaricus, Lactobacillus casei, Lactobacillus plantarum, Streptococcus faecium, and Streptococcus thermophilus, along with beneficial yeasts such as Torulopsis spp. and Aspergillus spp.
a) Mechanism of action Probios :
Production of lactic acid in the gut which reduce the pH and provides unfavorable conditions for pathogenic bacteria. Probios help in production of antibacterial compounds like lysozyme, lactoferrin, lactoperoxidase and bacteriocins. These compounds are bacteriostatic and bacteriocidal in nature. It reduces toxin production by suppressing the growth and colonization of E. coli. It increases Immunostimulation by increasing macrophage and lymphocyte activity. It rigorously competes with intestinal harmful microbes by competitive exclusion mechanism which help to restrict detrimental colonization to bind with receptors in the mucus layer. It helps in barrier function by improving mucin glycoproteins secretion through mucus producing cells to yield a dense mucus layer that helps to decrease intracellular permeability to pathogens.
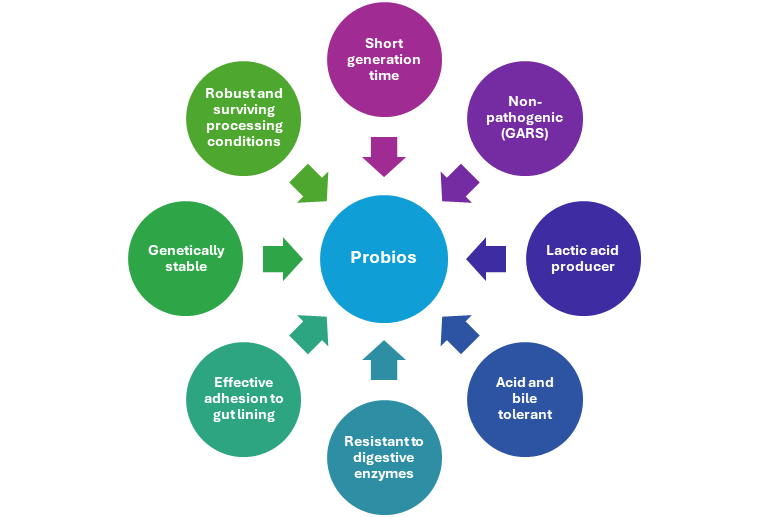
b) Charecteristic Of Probios
c) Benefits of Probios
- Minimize different kind of stress such as debeaking, vaccination and summer stress.
- Helps to maintain healthy gastrointestinal tract after antibiotic therapy.
- Reduce the incidents of chick mortality.
- Quicker detoxification of mycotoxins.
- Improves protein and fat synthesis.
- Improves enzymatic activity.
- Improves weight gain and FCR in broilers.
- Improves egg production, egg quality and shell quality in layers and breeders.
- Improves litter condition.
- Rapidly absorbed from the intestines to provide quick result.
- Very effective for mixed and gastro‑intestinal tract infections.
d) Comparision between Probios and common probiotic
| Probios | Common Probiotic |
| Made from direct fed microbial (DFM) | Made from spores |
| Contains 9 strains of microflora i.e. multistrain | Contains 2 to 3 species of microflora |
| Curdling of milk is observed when a teaspoon of Probios is added to milk, kept overnight | No curdling of milk is observed |
| High concentration of viable cells | Low concentration of viable cells |
| More viable in GI tract | Less viable in GI tract |
| Withstands pelletization temperature | Does not withstand pelletization temperature |
| Longer shelf life | Shorter shelf life |
9.Conclusion:
Probios, has demonstrated significant positive effects on poultry health and productivity. Its supplementation enhances gut enzyme activity, protein and fat metabolism, feed efficiency, fiber digestion, and organic phosphorus utilization, leading to improved body weight gain and feed conversion ratio in broilers. Probios also supports better litter conditions, reduces stress and mortality, promotes the development of intestinal mucous glands and villi, and maintains a healthy gastrointestinal tract. In layers and breeders, it improves egg production, egg quality, and shell strength. Additionally, Probios contributes to mycotoxin detoxification without causing adverse effects, making it a safe and effective probiotic solution for sustainable poultry production.
References
FAO, 2020, Gateway to poultry production and products. http:// www. fao. org/ poult ry- produ ction- produ cts/ produ ction/ poult ry- speci es/ en/. Accessed on June 11, 2020.
Garcia-Migura, L., Hendriksen, R. S., Fraile, L., & Aarestrup, F. M. (2014). Antimicrobial resistance of zoonotic and commensal bacteria in Europe: The missing link between consumption and resistance in veterinary medicine. Veterinary Microbiology, 170, 1–9.
Roth, N., Kasbohrer, A., Mayrhofer, S., Zitz, U., Hofacre, C., & Domig, K. J. (2019). The application of antibiotics in broiler production and the resulting antibiotic resistance in Escherichia coli: A global overview. Poultry Science, 98(4), 1791–1804.
WHO Factsheets. Antimicrobial resistance. April 2015. Available at: http:// www. who. int/ media centre/ facts heets/ fs194/ en. Accessed September 14, 2015.
Cui, S., Ge, B., Zheng, J., & Meng, J. (2005). Prevalence and antimicrobial resistance of Campylobacter spp. and Salmonella serovars in organic chickens from Maryland retail stores. Applied and Environmental Microbiology, 71, 4108–4111.
Diarrassouba, F., Diarra, M. S., Bach, S., Delaquis, P., Pritchard, J., Topp, E., & Skura, B. J. (2007). Antibiotic resistance and virulence genes in commensal Escherichia coli and Salmonella isolates from commercial broiler chicken farms. Journal of Food Protection, 70, 1316–1327.
Metchnikoff, E. (1907). Lactic acid as inhibiting intestinal putrefaction. In M. P. Chalmers (Ed.), The Prolongation of Life: Optimistic Studies (pp. 161–183).
Shokryazdan, P., FaselehJahromi, M., Liang, J. B., & Ho, Y. W. (2017a). Probiotics: From isolation to application. Journal of the American College of Nutrition, 36(8), 666–676. https:// doi. org/ 10. 1080/ 07315 724. 2017. 13375 29.
Shokryazdan, P., FaselehJahromi, M., Liang, J. B., Ramasamy, K., Sieo, C. C., & Ho, Y. W. (2017b). Effects of a Lactobacillus salivarius mixture on performance, intestinal health, and serum lipids of broiler chickens. PLoS ONE, 12(5), e0175959.
Food and Agriculture Organization/World Health Organization: “Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria.” Cordoba, Argentina: Author, 2001.
Chen, C. Y., Tsen, H. Y., Lin, C., Yu, B., & Chen, C. (2012). Oral administration of a combination of select lactic acid bacteria strains to reduce the Salmonella invasion and inflammation of broiler chicks. Poultry Science,91(9), 2139–2147.
Abushelaibi, A., Al-mahadin, S., El-tarabily, K., Shah, N. P., & Ayyash, M. (2017). Characterization of potential probiotic lactic acid bacteria isolatedfrom camel milk. LWT Food Science and Technology, 79, 316–325.
Lan, R., Tran, H., & Kim, I. (2017). Effects of probiotic supplementation in different nutrient density diets on growth performance, nutrient digestibility, blood profiles, faecal microflora and noxious gas emission in weaning pig. Journal Ofthe Science of Food and Agriculture, 97, 1335–1341.
Callaway, T. R, Edrington, T. S, Byrd, J., & Nisbet, D. J. (2017). Use of direct-fed microbials in layer hen production-performance response and Salmonella control, producing safe eggs: the microbial ecology of Salmonella. Pages 301−322 in Producing Safe Eggs. S. C. Ricke and R. K. Gast, eds. Academic Press, Cambridge, MA.
Gibson, G. R., Hutkins, R., Sanders, M. E., Prescott, S. L., Reimer, R. A., Salminen, S. J., Scott, K., Stanton, C., Swanson, K. S., Cani, P. D., Verbeke, K., & Reid, G.(2017). Expert consensus document: The international scientific association for probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nature Reviews Gastroenterology and Hepatology, 14, 491.
Food and Agriculture Organization of the United Nations/World Health Organization (FAO/WHO). Guidelines for the Evaluation of Probiotics in Food. In Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food; WHO: London, ON, Canada, 2002.
Salminen, S., Collado, M. C., Endo, A., Hill, C., Lebeer, S., Quigley, E. M. M., Sanders, M. E., Shamir, R., Swann, J. R., Szajewska, H., & Vinderola, G. (2021). The international scientific association of probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nature Reviews. Gastroenterology and Hepatology, 18, 649–667.
https:// doi. org/ 10. 1038/ s41575- 021- 00440-6.
Tortuero, F. Influence of the implantation of Lactobacillus acidophilus in chicks on the growth,
feed conversion, malabsorption of fats syndrome and intestinal flora. Poult. Sci. 1973, 52,
197-203.
Kabir, S.M.L.; Rahman, M.M.; Rahman, M.B. Potentiation of probiotics in promoting
microbiological meat quality of broilers. J. Bangladesh Soc. Agric. Sci. Technol. 2005, 2, 93-96.
Fuller, R. Probiotics in man and animals. J. Appl. Bacteriol. 1989, 66, 365-378.
Guillot, J.F. Les probiotiques en alimentation animale. Cah. Agric. 1998, 7, 49-54.
Fuller, R. The chicken gut microflora and probiotic supplements. J. Poult. Sci. 2001, 38, 189-196.
Liao, S. F., & Nyachoti, C. M. (2017). Using probiotics to improve swine gut health and nutrient utilization. Animal Nutrition, 3, 331–343. https:// doi. org/ 10. 1016/j. aninu. 2017. 06. 007.
Cotter, P. D., Ross, R. P., & Hill, C. (2013). Bacteriocins— a viable alternative to antibiotics? Natures Reviews Microbiology, 11, 95–105.
Grilli, E., Messina, M. R., Catelli, E., Morlacchini, M., & Piva, A. (2009). Pediocin A improves growth performance of broilers challenged with Clostridium perfringens. Poultry Science, 88, 2152–2158.
Rishi, P., Preet Singh, A., Garg, N., & Rishi, M. (2014). Evaluation of nisin-β-lactam antibiotics against clinical strains of Salmonella enterica serovar Typhi. Journal of Antibiotics, 67, 807–811.
Ricke, S. C. (2003). Perspectives on the use of organic acids and short chain fatty acids as antimicrobials. Poultry Science, 82, 632–639.
Ingram, L. O. (1989). Ethanol tolerance in bacteria. Critical Reviews in Biotechnology, 9, 305–319.
Bermudez-Brito, M., Plaza-Diaz, J., Munoz-Quezada, S., Gomez-lorente, C., & Gil, A. (2012). Probiotic mechanisms of action. Annals of Nutrition and Metabolism, 61, 160–174.
Collado, M. C., Gueimonde, M., Hernandez, M., Sanz, Y., & Salminen, S. (2005). Adhesion of selected Bifidobacterium strains to human intestinal mucus and the role of adhesion in entero pathogen exclusion. Journal of Food Protection, 68, 2672–2678.
Ayabe, T., Satchell, D. P., Wilson, C. L., Parks, W. C., Selsted, M. E., & Ouellette, A. J. (2000). Secretion of microbicidal alpha-defensins by intestinal Paneth cells in response to bacteria. Nature Immunology, 1, 113–118.
Callaway, T. R., Edrington, T. S., Anderson, R. C., Harvey, R. B., Genovese, K. J., Kennedy, C. N., Venn, D. W., & Nisbet, D. J. (2008). Probiotics, prebiotics, and competitive exclusion for prophylaxis against bacterial disease. Animal Health Research Reviews, 9(2), 217–225.
Hirn, J., Nurmi, E., Johansson, T., & Nuotio, L. (1992). Long-term experience with competitive exclusion and salmonellas in Finland. International Journal of Food Microbiology, 15, 281–285.
Penha Filho, R. A. C., Diaz, S. J. A., Fernando, F. S., Chang, Y. F., Andreatti Filho, R. L., & Junior, A. B. (2015). Immunomodulatory activity and control of Salmonella Enteritidis colonization in the intestinal tract of chickens by Lactobacillus based probiotic. Veterinary Immunology and Immunopathology, 167, 64–69.
Abbes, S., Ben Salah-Abbes, J., Jebali, R., Younes, B. R., & Oueslati, R. (2016). Interaction of aflatoxin B1 and fumonisin B1 in mice causes immunotoxicity and oxidative stress: Possible protective role using lactic acid bacteria. Journal of Immunotoxicology, 13, 46–54.
Han, J., Wang, Y., Song, D., Lu, Z., Dong, Z., Miao, H., Wang, W., He, J., & Li, A. (2018). Effects of Clostridium butyricum and Lactobacillus plantarum on growth performance, immune function, and volatile fatty acid level of caecal digesta in broilers. Food and Agricultural Immunology, 29,
797–807.
Aalaei, M., Khatibjoo, A., Zaghari, M., Taherpour, K., Akbari Gharaei, M., & Soltani, M. (2018). Comparison of single- and multi-strain probiotics effects on broiler breeder performance, egg production, egg quality and hatchability. British Poultry Science, 59(5), 531–538.
Sampath, V., Koo, D. H., Lim, C. B., & Kim, I. H. (2021). Supplemental effect of lactobacillus
plantarum on the growth performance, nutrient digestibility, gas emission, excreta microbiota, and meat quality in broilers. Brazilian Journal of Poultry Science, 23(04), 001–008.
Knap, I., Lund, B., Kehlet, A. B., Hofacre, C., & Mathis, G. (2010). Bacillus licheniformis prevents necrotic enteritis in broiler chickens. Avian Diseases., 54, 931–935.
De Los, G., Santos, J. R., Storch, O. B., & Gil-Turnes, C. (2005). Bacillus cereus var. toyoii and Saccharomyces boulardii increased feed efficiency in broilers infected with Salmonella enteritidis. British Poultry Science, 46(4), 494–497.
Beirao, B. C. B., Ingberman, M., Favaro, C., Jr., Mesa, D., Bittencourt, L. C., Fascina, V. B., & Caron, L. F. (2018). Effect of an Enterococcus faecium probiotic on specific IgA following live Salmonella Enteritidis vaccination of layer chickens. Avian Pathology, 47(3), 325–333.
Bailey, J. S., Stern, N. J., & Cox, N. A. (2000). Commercial field trial evaluation of mucosal starter culture to reduce Salmonella incidence in processed broiler carcasses. Journal Food Protection, 63(7), 867–870.
Kalavathy, R., Abdullah, N., Jalaludin, S., & Ho, Y. W. (2003). Effects of Lactobacillus cultures on growth performance, abdominal fat deposition, serum lipids and weight of organs of broiler chickens. British Poultry Science, 44(1), 139–144.
Yang, C. M., Cao, G. T., Ferket, P. R., Liu, T. T., Zhou, L., Zhang, L., Xiao, Y. P., & Chen, A. G. (2012). Effects of probiotic, Clostridium butyricum, on growth performance, immune function, and cecal microflora in broiler chickens. Poultry Science, 91, 2121–2129.






